- Are Shenzhen global digital marketing services suitable for export-oriented businessesMay 15, 2026View details
- What Are the Advantages of Shenzhen Global Digital Marketing ServicesMay 15, 2026View details
- Which internet marketing consulting company is better? First, look at the delivery logicMay 15, 2026View details
- How International Digital Agency Data Analysis Supports Ad Placement DecisionsMay 15, 2026View details
- Which Core Metrics Should You Focus on in Data Analysis for International Digital AgenciesMay 15, 2026View details
- Which industries are Guangzhou global digital marketing solutions suitable for?May 15, 2026View details
- What Are the New Trends in Guangzhou Global Digital Marketing SolutionsMay 15, 2026View details
- What problems can internet marketing consulting company services solveMay 15, 2026View details
New U.S. FDA regulations take effect on May 15: B2B official websites must embed a real-time compliance verification module
On May 15, 2026, the U.S. Food and Drug Administration (FDA) officially implemented supplementary provisions to the B2B Digital Compliance Statement Guidance. For the first time, this policy incorporates the digital compliance presentation capability of corporate official websites into the supply chain qualification assessment system, directly affecting the trade access efficiency and commercial trust foundation of Chinese exports of medical devices, food-contact materials, and consumer electronics products to the U.S.
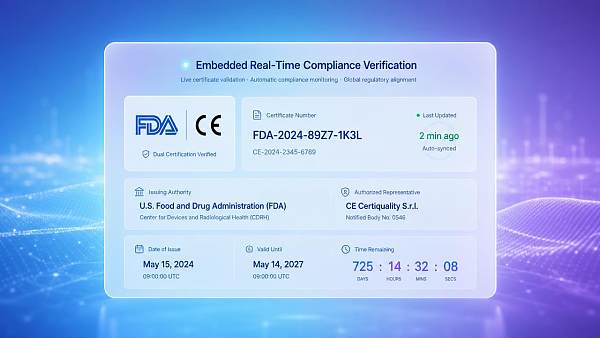
Event Overview
On May 15, 2026, the U.S. FDA officially implemented supplementary provisions to the B2B Digital Compliance Statement Guidance, requiring all Chinese suppliers exporting medical devices, food-contact materials, and consumer electronics products to the U.S. to integrate into their multilingual official websites a structured compliance statement module that can be verified online by the FDA (including CE/FDA dual-mark status, certificate number, issuing authority, and validity period), otherwise it will affect the pass rate of buyers' qualification pre-screening in the FDA Supplier Portal. This requirement directly affects overseas importers' supplier credibility assessment and order release decisions.
Which Segmented Industries Will Be Affected
Direct trading enterprises: As the registered entity in the FDA Supplier Portal and the signing party to procurement contracts, its official website is the primary source of information for importers conducting due diligence. If no verifiable module is embedded, it will lead to delays in qualification pre-screening or be flagged as “incomplete information”, significantly extending the onboarding cycle for new customers, and may trigger buyers to request additional costs such as third-party verification reports.
Raw material procurement enterprises: Although they do not directly export finished products, the key raw materials they supply to downstream manufacturers (such as medical-grade silicone, food-grade PC resin, and RoHS-compliant chips) must be accompanied by traceable compliance certificates. The new regulation forces their official websites to simultaneously upgrade statement modules to support downstream customers in fulfilling complete machine/component compliance disclosure obligations, otherwise they face the risk of weakened supplier relationships.
Processing and manufacturing enterprises: Enterprises undertaking OEM/ODM roles often apply for FDA listing in the brand owner's name, while their own official websites have long lacked independent compliance identification. After implementation of the new regulation, importers will be more inclined to choose manufacturers with independent digital compliance infrastructure capabilities to reduce joint liability risks; those lacking this capability may be excluded from preferred procurement lists.
Supply chain service enterprises: Including third-party service providers such as compliance consulting, certification agency services, and digital platform development, whose business model is shifting from “one-time certificate services” to “continuous compliance data operations and maintenance”. The new regulation objectively accelerates the market penetration of compliance SaaS tools, while also imposing higher requirements on service providers' data structuring capabilities, API integration experience, and multilingual content management.
Key Points of Attention and Response Measures for Relevant Enterprises or Practitioners
Confirm whether the official website meets structured data specifications
It is necessary to verify whether the official website adopts the Product and Certification extension fields in the Schema.org standard, supporting the FDA system in automatically capturing core parameters such as certificate number, issuance date, validity period, and certification body URI, rather than merely displaying static PDF scans.
Clarify the consistency of compliance statements across multilingual pages
Both the English main site and the Chinese sub-site must deploy verification modules with equivalent effect; if only the English pages are embedded while the Chinese pages are missing them, the FDA system may determine this as “information asymmetry”, affecting the overall credibility score.
Establish a dynamic certificate update mechanism
The module must achieve lightweight integration with the internal certificate management system (such as QMS) or the certification body database, ensuring that when certificates expire, change, or are revoked, the official website statement is synchronously refreshed, avoiding buyer misjudgment caused by outdated information.
Reserve a testing window for the FDA verification interface
It is recommended to complete module invocation testing in the FDA Supplier Portal sandbox environment before the third quarter of 2026, and obtain official returned verification log samples for internal technical calibration and legal review.
Editorial Viewpoint / Industry Observation
Observably, this regulation is not merely a technical requirement but a strategic signal: FDA is shifting from document-centric to data-centric oversight in global supply chains. Analysis shows that the real impact lies less in upfront development cost and more in operational discipline — firms with fragmented IT systems or siloed compliance functions face higher integration friction. From industry perspective, the move accelerates the convergence of regulatory compliance, digital marketing, and procurement trust infrastructure. It is better understood as a de facto standardization push for B2B transparency, rather than an isolated audit trigger.
Conclusion
This new regulation marks that regulatory compliance is moving from “post-event inspection” toward “in-process visibility”, posing a substantive test to enterprises' digital infrastructure capabilities. Its long-term significance lies not in adding another market access barrier, but in driving Chinese export enterprises to upgrade compliance from a legal support function into a digital asset that runs through the entire product lifecycle. Rationally speaking, enterprises that take the lead in completing module deployment may gain hidden competitive advantages such as priority buyer ratings, shortened factory audit cycles, and reduced evidentiary costs in quality disputes.
Information Source Description
Based on the Guidance for Industry: Supplemental Requirements for B2B Digital Compliance Statements published on the official U.S. FDA website (publication date: November 20, 2025, FR Doc. No. FDA-2025-D-18742); with simultaneous reference to FDA Supplier Portal technical documentation v3.2 (updated version in April 2026). It is necessary to continue monitoring whether the FDA will subsequently issue a module compatibility whitelist, detailed exemption rules for SMEs, and transitional enforcement standards.
Related Articles
Related Products